Fast-tracked
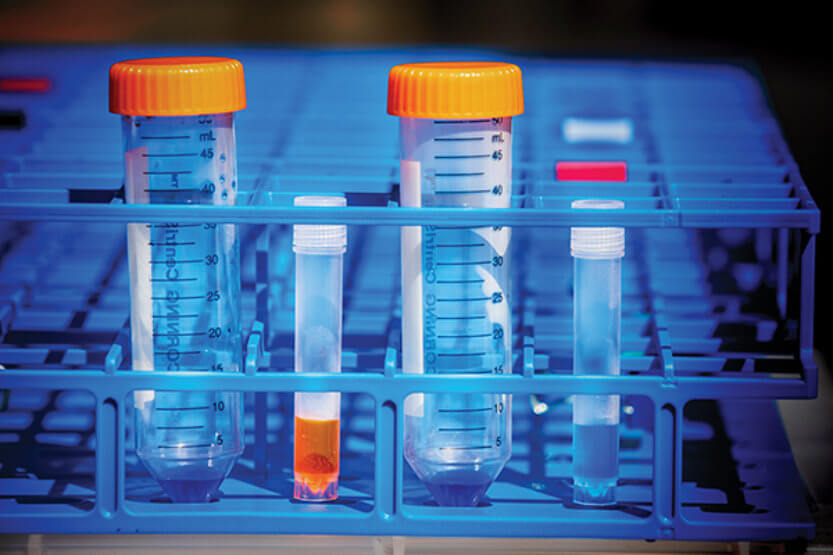 The University transitioned to smaller tubes for saliva collection for their campus-wide COVID-19 testing program. The smaller tube can be opened in the lab by robotics, expediting test result turn-around time. (Image by Fred Zwicky)
The University transitioned to smaller tubes for saliva collection for their campus-wide COVID-19 testing program. The smaller tube can be opened in the lab by robotics, expediting test result turn-around time. (Image by Fred Zwicky) “The pandemic was bearing down on us, and we knew we had to do something,” recalls Martin Burke, associate dean of research at the Carle Illinois College of Medicine. Burke had just been asked by Andreas Cangellaris, vice chancellor for academic affairs and provost, to assemble a team to develop a plan that would make it safe for students to return to campus for the fall semester. The first person Burke contacted was Paul Hergenrother, professor of chemistry at the Carl R. Woese Institute of Genomic Biology. A friend and colleague of Burke’s dating back to their post-graduate work at Harvard two decades earlier, Hergenrother was already mulling the most critical piece of that enormous challenge—devising a testing procedure rapid enough to control the spread of COVID-19 in a community of 50,000 people working and living in close quarters.
Hergenrother was certain of one thing—nasal-swab testing, the most widely used method at the time, was not an option. Labs nationwide were experiencing critical shortages of the supplies needed to conduct such tests. “Even health-care workers couldn’t get tested then,” he says.
Meanwhile, Dr. Diana Ranoa, MS ’08 LAS, PHD ’13 LAS, a postdoctoral researcher in Hergenrother’s lab, had alerted him to research conducted at Yale showing that the COVID virus could be detected in saliva, as well as in nasal samples. On a Sunday evening video call in early May, Hergenrother organized a brainstorming group that included Ranoa, two other post-docs and a grad student; they hatched a plan to launch what he calls “a Manhattan Project-style effort,” a reference to the University of Chicago scientists who engineered the first controlled, self-sustained nuclear chain reaction in 1942. The goal for Hergenrother and his team was to start saliva pilot testing by early July. “We got to work the next morning,” he says.
The first step required confirming the viability and accuracy of saliva testing, which they did by using 100 samples collected from COVID-19 positive patients at Carle Foundation Hospital, a key partner, along with the Champaign Urbana Public Health District, in a local pandemic control effort. But even saliva testing involves a series of steps that require isolating the RNA molecule so that it can be converted to DNA, which then must undergo a chemical process called a polymerase chain reaction (PCR) to amplify the DNA and make it measurable.
Hergenrother and his group knew those procedures would be far too laborious once the campus reopened, and at least 10,000 people would need to be tested each day. They experimented with a matrix of conditions—time, temperature, additives—and determined that it was possible to bypass the need for RNA isolation and go directly to PCR diagnosis by heating saliva samples at 95 degrees Celsius (203 degrees Fahrenheit) for half an hour.
“We were the first ones to show that you could do this from saliva, which was very significant,” Hergenrother says. Another significant benefit was that the new procedure inactivated the virus itself, making the entire process safer for the lab technologists and health-care workers who would handle the samples.
The next challenge was regulatory: The group had to submit their findings to the Food and Drug Administration for Emergency Use Authorization, which would allow use of the test. (It was approved.) Hergenrother says their research manuscript, available online, now serves as “a blueprint” for other labs to follow.
Multifaceted initiative
Burke says Hergenrother’s work was a “key breakthrough,” but only one piece of the multifaceted initiative he’s dubbed SHIELD, which signifies the effort to “shield” the U of I campus and Champaign-Urbana community from COVID-19. Rapid saliva testing could only work if there was a laboratory on campus large enough to accommodate the enormous volume of samples that would need to be evaluated. For that, Burke and Hergenrother enlisted the help of Professor Tim Fan, PHD ’07 VM, an oncology specialist in the College of Veterinary Medicine, who thought it would be possible to convert a section of the Veterinary Diagnostic Laboratory into a dedicated COVID testing facility.
“That required a Herculean effort, with a lot of regulatory hurdles to clear,” says Hergenrother, explaining that every diagnostic laboratory has to qualify for federal certification. “Tim and his team did all of that while we were developing the test, including ordering all the necessary equipment.”
By July 6, the testing operation was up and running with a pilot program to test faculty, staff and students who were on campus over the summer. Burke assembled a team of more than 300 people to ramp up the effort in preparation for the impending arrival of undergraduates just a few short weeks away. At the converted vet diagnostic lab, Ranoa partnered with veterinary postdoctoral researcher Robin Holland, PHD ’18 VM, DVM ’20, to train additional lab technicians for round-the-clock testing, done in three eight-hour shifts. “Our assembly line requires that everyone perform their job with the utmost accuracy,” Ranoa says. “One small error at any step of the process can have profound effects on at least one individual at the receiving end. We are transitioning to a robotic system that will help ease the pressure.”
In addition, numerous “little innovations had to be made along the way,” Hergenrother says. Initial supplies of test tubes had to be sterilized and capped by hand, so he and a cohort of graduate students gathered one evening to clean and cap thousands of tubes. When new, smaller sterile tubes that were compatible with robotics became available, Beckman Institute Director Jeff Moore, ’84 LAS, PHD ’89 ENG, and a student team “devised and constructed a simple, ingenious rack system” that allowed an entire rack of tubes to be placed into the heating bath as soon as saliva samples arrived at the lab.
When students returned in late August, they encountered some curious new installations located throughout the campus—17 tents housing 40 saliva-sample collection stations for mandatory COVID testing. Students were required to submit samples twice a week, while faculty and staff were tested a week, since undergraduate positivity rates were higher during pilot testing. Students also discovered new COVID-related security rules for gaining access to campus buildings and an innovative app for learning their test results (see sidebar). Hergenrother says that samples are picked up and driven to the diagnostic lab hourly, and samples from those who are experiencing symptoms or believes they have been exposed to COVID-19 are placed in a “priority rack” for immediate processing. The lab has processed as many as 18,000 samples in one day, and the system’s goal is for anyone testing positive to be notified within 30 minutes of their test result so they can isolate and receive medical treatment. Test results are typically available within two to six hours.
Shortly after the fall semester began, the school received unfavorable national publicity when off-campus parties led to a COVID outbreak, spiking the positivity rate above 2 percent. Burke and Hergenrother are emphatic in saying the incidents represented only a small minority of students. The SHIELD system made it possible to promptly identify and isolate the infected students, and the student testing requirement was increased to three times a week. The positivity rate was reduced to well under 1 percent in a matter of a few days—evidence, Burke says, that the system is working.
Illinois Governor J.B. Pritzker has repeatedly praised the U of I’s efforts in his daily COVID press briefings, and Burke says that U of I System President Tim Killeen has called it “an Illinois moment.” The SHIELD system also is in use at UIC and UIS, and the University has made it available to institutions throughout the state. In August, the U of I announced the formation of a University-related organization designed to make the program available nationally.
Both Burke and Hergenrother make liberal use of the term “awesome” to describe the efforts of SHIELD team members and the University’s many interdisciplinary collaborations.
“We didn’t realize it at the time, but for about four years prior to the pandemic, we were getting prepared for it,” Burke says. He notes that the establishment in 2015 of the Carle Illinois College of Medicine with its stated mission—medical education through the lens of engineering—“was the major catalyst that allowed us to be ready because of the bridges and partnerships we had formed across the entire campus. When COVID struck, we were able to respond with the most interdisciplinary, interconnected network you could imagine. This is interdisciplinary on steroids. We’re not celebrating anything yet, and in the meantime, we’re not letting up.”

The Safer Illinois app offers several dynamic health-related features. (Image courtesy of Safer Illinois)
Quick and easy
Mobile phone app Safer Illinois helps students, staff and faculty track their COVID-19 test results and more
In an effort to control the spread of COVID-19 at the start of the fall semester, the University began requiring all students, faculty and staff to have a current negative COVID test result to gain entry into any campus building. The campus provided two options to make it possible to comply with the directive: a website called Boarding Pass, which links a person’s COVID testing status to the University ID system; and a free, downloadable mobile phone app called Safer Illinois, which runs on IOS and Android platforms.
Professor Martin Burke, SHIELD team leader, says the Safer Illinois app is “very strongly recommended” for all faculty, staff and students because it offers several dynamic health-related features. These include reporting of COVID-19 test results, reminders about test schedules, links to online health care and even notification of possible coronavirus exposure.
The Safer Illinois app grew out of an interdisciplinary program called ROKWIRE, a University initiative launched in 2018 to create an open-source software platform that could serve as a single entry point for the entire U of I community. The first Rokwire app provided information about dining, events and athletics. ROKWIRE is headed up by Landscape Architecture Professor William Sullivan, ’80 ACES, MS ’82 ACES.—P.E.



